- Alternative balanced equation for the cracking of decane cracked#
- Alternative balanced equation for the cracking of decane Crack Serial#
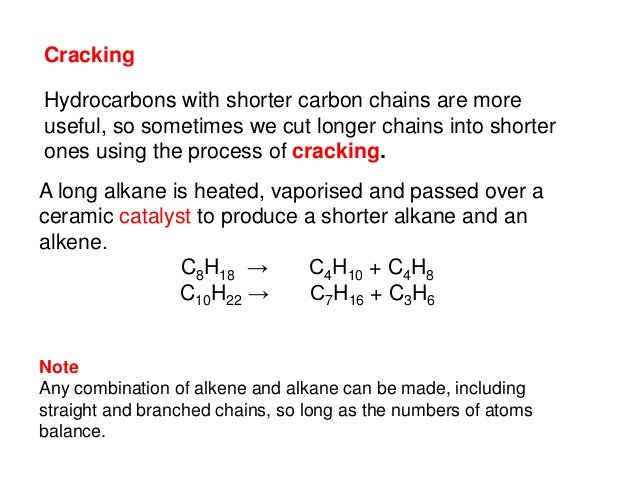
The process for steam cracking is essentially the same. One of the hydrocarbons produced will be an alkane and the other will be an alkene. Thermal decomposition occurs as the longer chained hydrocarbon splits up into smaller chained hydrocarbons.Pass the vaporised long-chained hydrocarbon over a powdered catalyst sometimes aluminium oxide (a catalyst is something that increases the rate of reaction without being used up in the reaction).Heat up the long-chained hydrocarbon so that they turn into a gas (vaporise).There are 2 different types of cracking catalytic cracking and steam cracking. Some of the smaller hydrocarbons are used as fuels and others are used to make polymers.Ĭracking is a thermal decomposition reaction, which is the breaking down of molecules using heat (thermal decomposition is an endothermic reaction as energy/ heat is taken in). Cracking is a process that breaks down less useful longer chained hydrocarbons, like bitumen, into shorter more useful hydrocarbons, which are less viscous and easier to ignite. One of the by-products of this thermal decomposition reaction is ethene.Shorter chained hydrocarbons are more useful than longer chained ones because longer chained hydrocarbons are very viscous (do not flow well – like golden syrup) and are harder to ignite (set fire to). Summary Oil companies break large hydrocabon molecules down into smaller, more useful hydrocarbons.Ĭracking takes place at high temperatures as the large molecules pass over a catalyst. They are less likely to clog up your arteries with the fatty deposits that can cause heart disease.
Alternative balanced equation for the cracking of decane Crack Serial#
Decane Ing Equation Serial Generator 1īackuptrans iphone whatsapp transfer keygen crack serial generator 1. They are a healthier option than the saturated fats we eat in butter. Some of them are called polyunsaturates because their molecules have more than one double bond. Have you heard of the unsaturated oils we use to make margarine These contain double bonds in their carbon chains too.
So while the alkanes are saturated hydrocarbons, the alkenes are unsaturated hydrocarbons. Lets look at the structures of the first three members of the alkenes, shown in Fig.2 below.ĪtomAn atom is the smallest particle of an element that can still be defined as that element. C10H22 (g) - C8H18 (g) + C2H4 (g) Organic compounds Crude oil is a mixture of hydrocarbons and is separated into.
Alternative balanced equation for the cracking of decane cracked#
For example, decane (an alkane with 10 carbons) can be cracked to produce octane and ethene. Its name is ethene and it is the smallest member of the family of hydrocarbons called the alkenes. The catalyst works by providing the hydrocarbon gases with a convenient surface for the cracking to take place. Molecule formed in the equation given for cracking in the previous section Heres the equation again: C 10H 22(g) C 8H 18(g) C 2H 4(g) Remember that the general formula for an alkane is C nH (2n2). MoleculeA molecule is a group of two or more atoms bonded together. The fractions that provide us with fuel for cars are in great demand.īut we just dont use so much of the heavier fractions, so chemists have found ways to meet the demand for petrol by producing this from the heavier hydrocarbons.ĬatalystA catalyst is a substance that alters (usually speeds up) the rate of a chemical reaction, but remains chemically unchanged itself at the end of the reaction.Ĭatalyst, and they break down into smaller molecules.Ī catalyst is a substance that speeds up a chemical reaction but remains chemically unchanged itself at the end of the reaction.) Look at an example of cracking below: C 10H 22(g) C 8H 18(g) C 2H 4(g) Cracking is an example of a thermal decomposition reaction (in this case of an. Write a balanced symbol equation for this reaction.Ĭrude oilCrude oil is a liquid formed from tiny sea creatures and plants that died millions of years ago.Īs you might expect, we do not need as much of some fractions as others. Explain the meaning of the words underlined.Ĭracking decane can produce a mixture of butane and propene in the ratio 1:2 respectively.
